Electroconvection in Smectic Liquid Crystal Films


If you apply a voltage across a fluid it will convect if it can develop some unbalanced space charge which is pushed on by the applied field. In typical electrolytes, one has a lot of ions of both signs and any applied DC field is quickly screened out by the formation of charged layers on the electrodes, leading to "electroneutrality" (i.e. no space charge) in the bulk of the fluid, and hence no electroconvection. Unbalanced charge layers can also form at free surfaces, however, and forces on these can cause convection. In our experiments, we use a film of fluid which is nearly all surface: a suspended smectic liquid crystal.
Smectics have molecules that like to lie in layers and easily form very thin suspended films, much like soap bubbles, except without the solvent. In fact, "smectic" just means "soapy". The odd thing about smectic films is that they can flow in the plane of the layers, but very strongly resist making new layers, so they can flow without changing thickness. You can make a film 1 mm wide that flows like crazy, but is only 3 molecules thick. It makes a nearly perfect two-dimensional fluid.
To make smectic films electroconvect, we suspend them between (say) two wires and apply a voltage across the wires. Below the onset of convection, the configuration of charges and fields which develops looks like this in cross section:
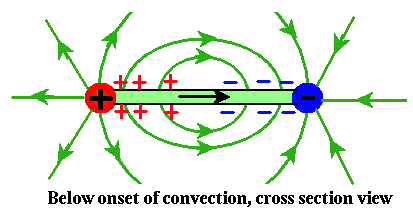
The green rectangle is the film, see edge on. Actually, it is about a 100,000 times thinner than shown here. The wires stick out of the screen. The green arrows show the electric field outside the film. The upshot is, a bunch of positive charge develops on the surfaces near the positive electrode, and a bunch of negative charge near the negative electrode. The black arrow shows the direction of current flow and the electric field inside the film. This field pushes on the surface charge layers, which tends to destabilize the fluid to convection. In top view, below the onset of convection:
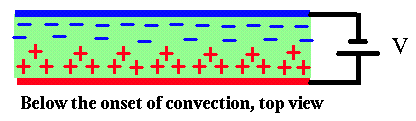
The situation is much like in Bénard convection, in which the lighter fluid is on the bottom and the heavier one on top: here the charges are nearer the electrodes of the same sign. If the voltage is small, the film just carries the current by conduction and does not move. However, if the voltage exceeds a threshold value (which is about 10 volts in our experiments) the electrical forces lead to an instability and the films starts flowing in a series of vortices like this:

Above onset, the film carries a bit more current because some is transported by the moving fluid. The vortices are a nonlinear solution of the fluid equations; they are the "pattern" that we are interested in.
Another nice thing about electroconvection, unlike Bénard convection, which depends on gravity, is that we can wrap the film around on itself and eliminate the ends. Schematically, an annular film looks like:
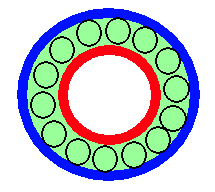
We are currently working on the annular version of this experiment with the extra feature that one can independently apply a shear by rotating the inner electrode. For various papers on this, both experimental and theoretical, see the smectic papers page.


The Experimental Nonlinear Physics Group, Dept. of Physics, University of Toronto,