![]() PHY385 Module 3
PHY385 Module 3
Student Guide
Concepts of this module
Irradiance
Radiation pressure
and momentum. Index of reflection.
Dispersion in
dielectrics
Quiz
![]() Activity 1 - Irradiance
Activity 1 - Irradiance
Solving theoretical problems, suggested by the
Practicals Instructor, study the relationship among irradiance, flux
density, Poynting vector and energy volume density for electromagnetic wave.
![]() Activity 2 - Radiation pressure and
momentum. Index of
reflection.
Activity 2 - Radiation pressure and
momentum. Index of
reflection.
For this Activity, you need to know the definition of
the index of reflection and the properties of a photon. The index
of reflection of a surface is given by R = ureflected/
uincident and shows the fraction of
the incident energy being reflected by the surface. The photon energy is E = fh; and
the photon momentum is p = E / c, where c is the speed of light in free space.
1. Find the total linear momentum
transferred to the completely absorbing screen that receives 300 W of light for
100 s.
2. What is the
momentum of a 1019 - Hz X-ray photon?

Activity 3 - Dispersion in
dielectrics
The dispersion is a phenomenon of dependence of the
index of refraction of the monochromatic wave (or the phase speed of
propagation of the wave) in a substance on the wavelength (see Lecture 6).
The textbook suggests a linear function for the
relationship between the refractive index of a medium and the wavelength:
![]() (see
page 70, Chapter 3).
(see
page 70, Chapter 3).
Lecture 6
contains a diagram for estimated behaviour of the function n(λ) for liquids and solids.
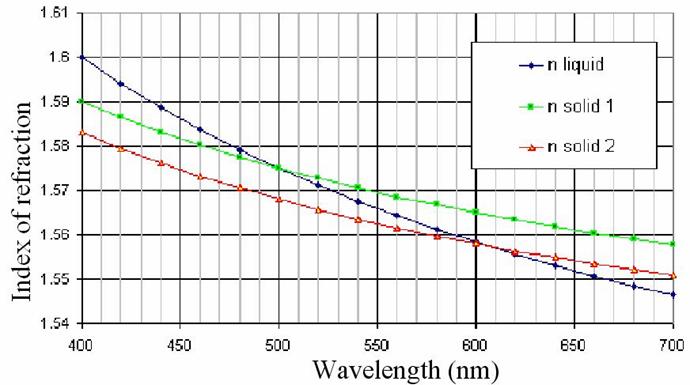
Assume that air has n = 1. With water and glycerine samples prove either of the above relationships.
For the experiment you will use the following
equipment:
- three plastic cups with a washer at the bottom;
- a vessel with tap water;
- a vessel with glycerine;
- a ruler;
- a set of filters transparent for different wavelengths: 436
nm, 520 nm, 620 nm and 675 nm; and
- tissues for keeping your workplace, equipment and your hands
dry and clean.
Place two empty cups close to each other and look at
the washer from the top positioning your eye closer to the top of the cup. With
the ruler outside the cup define the distance from your eye to the washer and
record the value. If all lab partners perform this measurement in turn, the
average value will account for the random uncertainty.
I. Experiment with water.
1. Time all steps of
the experiment with water to know whether you will have time to repeat
measurements with glycerine.
2. Fill one of the
cups with water to the top. The deeper the liquid, the more accurate
measurements are expected. However, you should be careful working with liquids
in such unstable vessels!
3. Look at the washer
normally to the water surface positioning your eye close to the water surface.
With the ruler outside the cup, measure the distance to the image of the washer
and record this measurement.
4. The index of refraction
of white light in water is about 1.3333. Using your measurements, compare the
experiment result with the expected value of 1.3333 and give a difference in
percents.
5. Repeat the
experiment with at least three filters: blue, green and red. Calculate the
index of refraction of specific wavelengths in water. It is recommended that
you save 5 significant figures in the value of n. Plot the results of measurements either as "n vs.
λ" or as "(n2 -1)-1 vs. λ-2
".
6. Conclude on the
dispersion relation of the refractive index in water.
7. Put the cup with
water aside, far from your workplace.
II. Experiment with glycerine. (Do this
experiment only if your records show that there is enough time before the end
of the session).
1. Use the third cup
with a washer to perform all steps explained above with the glycerine
99%-solution. The expected value for white light is 1.4723.
2. Fill the cup with
glycerine almost to the top. The deeper the liquid, the more accurate
measurements are expected. However, you should be careful working with liquids
in such unstable vessels!
3. Put the cup with
glycerine on the lab table far from your workplace and clean the table you are
working at.
4. Conclude on the
accuracy of the results for the used substance(s) and the sources of
uncertainty.
This Student Guide was created by Natalia
Krasnopolskaia in September 2014.